Rowan and Thor were interviewed on the Stem Cell Podcast to discuss our new paper on human blastoids cultured to post-implantation stages. Check out the episode via this link:
The Stem Cell Podcast


Rowan and Thor were interviewed on the Stem Cell Podcast to discuss our new paper on human blastoids cultured to post-implantation stages. Check out the episode via this link:
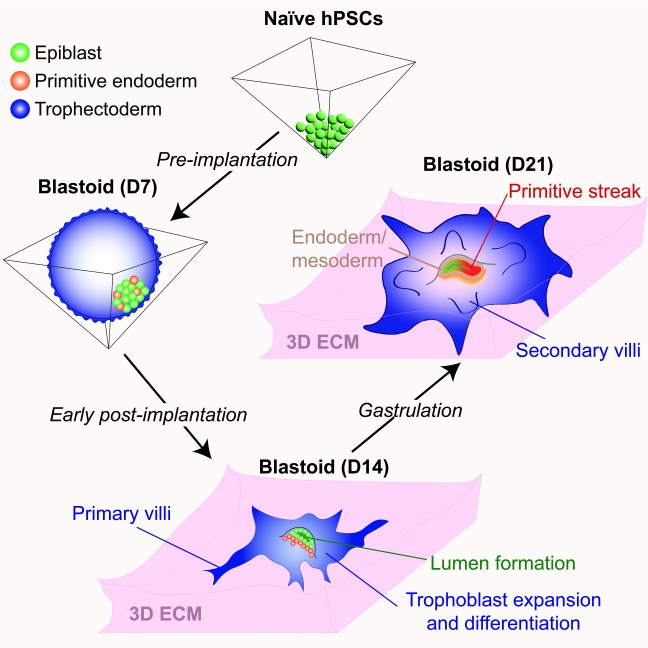
Our paper on extended human blastoid culture was published today in Cell Stem Cell. In a transformative advance, several groups have reported that naive human pluripotent stem cells (hPSCs) can form blastocyst-like structures (also known as “blastoids”) that model the human pre-implantation embryo. However, the extent to which blastoids can recapitulate defining features of post-implantation development remained unexplored. In this study, we optimized the conditions for blastoid generation from naïve hPSCs and investigated their capacity for extended culture on thick 3D extracellular matrices, which better mimic the physical environment of the human endometrium compared to flat surfaces. We developed an experimental methodology that supports human blastoid culture for up to day 21 (D21), including the formation of complex embryonic and placental structures. By performing a detailed single cell transcriptome analysis at three distinct time points (D7, D14, and D21), we benchmarked our model system to human embryos at pre-implantation, early post-implantation, and early gastrulation stages.
3D-cultured human blastoids display several molecular and morphogenetic hallmarks of early post-implantation development, including lumenogenesis of the epiblast compartment, rapid expansion and diversification of trophoblast lineages, and robust invasion of extravillous trophoblast cells by D14. Extended blastoid culture resulted in the formation of a primitive streak-like structure, as evidenced by the localized activation of TBXT (Brachyury) by D18. Blastoids maintained until D21 acquired a single cell transcriptome profile that closely resembled that of a gastrulating human embryo analyzed at Carnegie Stage 7. This included the emergence of blastoid primordial germ cells, definitive endoderm, and various mesodermal lineages, and a diverse array of extraembryonic cell types, including blastoid amnion, cytotrophoblast, extravillous trophoblast, extraembryonic mesoderm, syncytiotrophoblast, and yolk sac endoderm. Thus, the 3D-cultured human blastoids described herein model embryonic and extraembryonic development from pre-implantation to early gastrulation stages, offering a continuous and integrated in vitro model system of early embryogenesis.
Congratulations to Rowan, our collaborators in the Dietmann and Zhou labs, and the entire team!
Note: The generation of integrated models of human development in our laboratory is entirely supported by private foundation grants and does not involve federal funding from the National Institutes of Health.

We recently welcomed two new graduate students to our lab:
Kyoung Park earned a Master of Science in Biology from Yonsei University, South Korea, and previously served as a Research Technician in our lab from 2018 to 2022. She played a key role in starting the lab and collaborated closely with Dr. Jianlong Wang’s lab at Columbia University on the OCT4 protein interaction network in naive and primed hPSCs, identifying pluripotent-state-specific interactions with distinct subunits of the BAF chromatin remodeling complex. She enrolled in the Developmental, Regenerative, and Stem Cell Biology Program in 2022 and re-joined our lab as a graduate student in March 2023.
Richard Yin is a member of the Developmental, Regenerative, and Stem Cell Biology Program and joined the Theunissen lab in August 2023. He earned his B.S. from the University of Toronto in 2022. He started his research journey as a volunteer in Dr. Phedias Diamandis’ lab, studying potential therapeutic targets in glioblastoma tumour microenvironments. As an undergraduate researcher under Dr. Jennifer A. Mitchell, he investigated the evolutionary role of human-specific KLF4 enhancer candidates in pluripotent stem cells. His passion for stem cell research led him to pursue a Ph.D. at WashU. Outside the lab, he enjoys cooking, rock-climbing, gaming, and movies.
Welcome Richard and welcome back Kyoung!
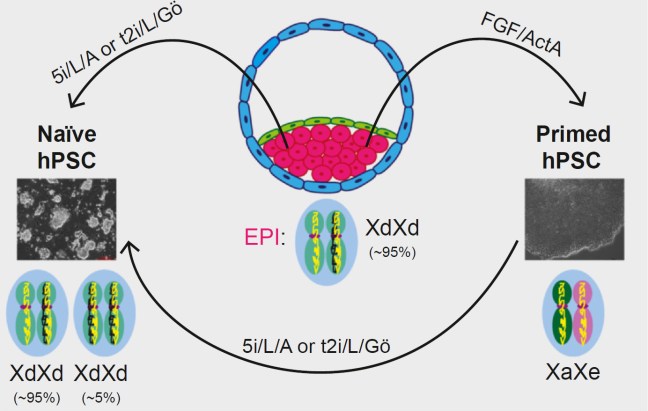
Our review on stem-cell-based models of X-chromosome inactivation and reactivation during human development was published in the Special Issue on Early Embryonic Development Models in Current Opinion in Genetics & Development. Congrats Shafqat!

Rowan’s ISSCR talk on extended human blastoid culture was featured on the latest episode of The Stem Cell Podcast. Thanks Drs. Daylon James and Arun Sharma for your positive appraisal of our work! You can listen to the podcast using the link below (Rowan’s talk is discussed from 13:30 mins onwards…):